The oral microbiome is a collection of microbes living in our mouths. They have a significant impact on our overall health. Periodontitis can be caused by bacteria, which can also lead to other systemic diseases like diabetes and heart disease. Some types of cancer have also been linked to other oral organisms. Scientists are studying how these microbes interact with each other and our bodies to discover their roles in health and disease.
One group that belongs to Candidate Phyla Radiation is one of the many bacterial species found in our mouths. These bugs are particularly mysterious because they are extremely small and live in a unique symbiotic relationship with their host bacteria. Only one group of bacteria from the CPR has been studied in depth, a group called TM7. This group was first cultivated by Dr. Xuesong He at Forsyth Institute in 2014.
Dr. Dr. Jeffrey S. McLean, a University of Washington collaborator, has developed a new model system that uses the first human-derived TM7 strain, TM7x. It also includes its host bacterium Actinomyces odontolyticus. The model system was used to experimentally study these tiny bacteria. It allowed researchers to test a hypothesis on how TM7 adapts to live inside humans and provided empirical evidence to support previous genomic studies. Today, their findings were published in the journal. Proceedings of the National Academy of Sciences.
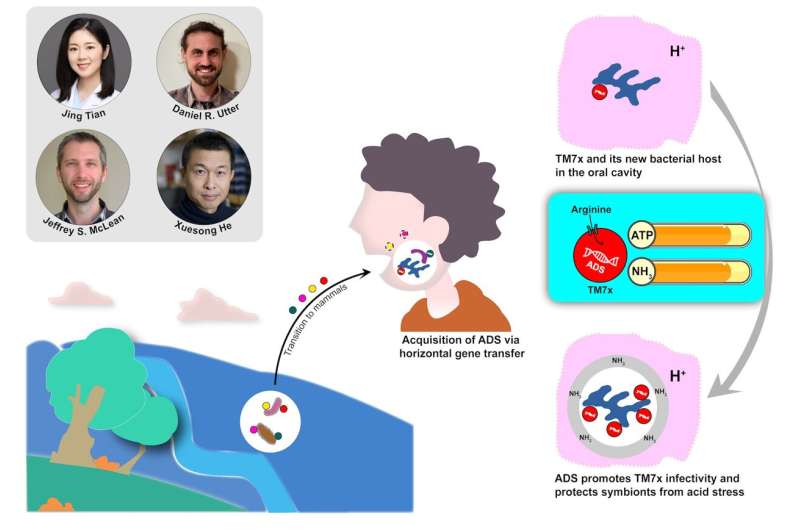
Scientists have found TM7 within many environments, including soil, groundwater, as well as the bodies of other mammals. Studies have shown that although the genome is very similar overall, the TM7 in the human mouth is unique because it contains a gene cluster encoding ADS.
Dr. McLean said, “This was interesting to us because there seem very few genomic modifications that occurred in this small group of bacteria with already small genomes when they transitioned form the environment to mammals.”
Researchers believe that TM7 acquired ADS in an evolutionary advantage to adapt and survive in human oral cavities. Dr. Jing Tian (first author of the study) used the model system as a laboratory to test the hypothesis. ADS helped TM7x to break down arginine. This process produces the compounds Adenosinetriphosphate (ATP), as well as ammonia. TM7x’s infectivity, or ability of to multiply, was boosted by the increased levels of ATP/ammonia. It protected TM7x as well as its host bacterium against acid stress, a condition microbes often encounter in the human oral cavity because of the acid that is created when bacteria metabolize and eat dietary carbohydrates.
The experiment proved that TM7x survived in the experimental environment for longer periods of time than they could without ADS.
“CPR bacteria studies are based on a culture independent genomic approach. Dr. He.
“The production ammonia by TM7 encoded ADS raises pH in the human dental microenvironment,” said Dr. Tian. He is a Pediatric Dentist at Peking University Hospital and Stomatology, and a visiting scholar at Forsyth. Dr. Tian previously found that children with dental caries had a significantly higher abundance of TM7 after they were treated. Dr. Tian stated that this suggests that TM7 might be more closely associated with a state free from caries. He plans to continue his research in this area.
This study adds to a growing body evidence that TM7 bacteria may play a greater protective role in oral hygiene than originally thought. Scientists believe that TM7 bacteria is responsible for periodontal disease. For example, TM7 levels are found to be high in patients with periodontal disease. A recent study led by Dr. Batbileg Bor of Forsyth found the opposite effectTM7 reduced periodontal inflammation, bone loss, and bone loss in a mouse model.
Dr. McLean stated that “we are still in the early stages understanding how each one of these ultrasmall parasitic bacterias, which we basically just discovered within humans, are impacting on health and disease.”
Dr. He agreed. “Now, for TM7, we have a manipulatable modeling system, which is a huge advantage.”
Groundwater, moose, and other groundwaters are home to ultra-small parasitic bacteria.
An earlier study: Otari Chipashvili and colleagues, Episymbiotic Saccharibacteria suppresses gingival inflammation in mice and bone loss through host bacterial modulation. Cell Host & microbe (2021). DOI: 10.1016/j.chom.2021.09.009
The acquisition of the arginine-deiminase system benefits epiparasitic Saccharibacteria as well as their host bacteria in a mammalian niche. Proceedings of National Academy of Sciences (2022). DOI: 10.1073/pnas.2114909119.
Citation:
Researchers gain insight into how ultrasmall organisms from the environment have evolved to live inside people (2022, January 3).
Retrieved 3 January 2022
from https://phys.org/news/2022-01-gain-insights-ultrasmall-bacteria-environment.html
This document is subject copyright. Except for fair dealings for private study or research purposes, there is no
Without permission, part may be reproduced. The information is provided only for informational purposes.

